 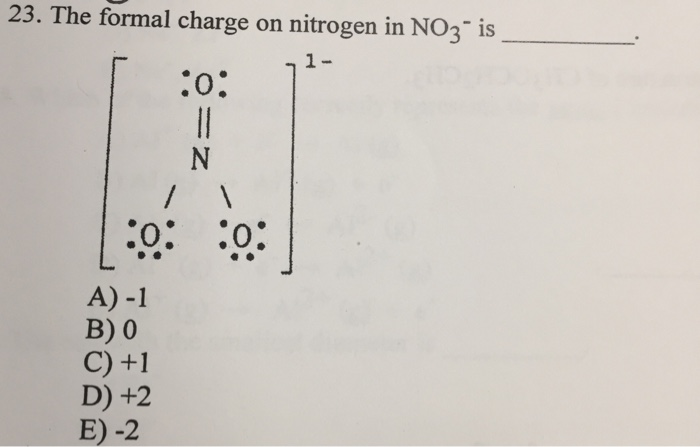 
The nitrogen in the center is Always positive, and the three oxygen atoms have one bond to nitrogen, and then a dashed bond. Go ahead and look at the Lewis diagram of Nitrate. The reason is not just formal charges but also resonance hybrid stabilization. This is just the entirety of the molecule, but why? and how does nitrogen in this molecule contain a positive charge, but its only bonded to 3 things and the overall molecule is negative? Nitrate has an overall charge to the entire molecule, of -1. Now lets take this a step further and talk about Nitrate. There is a positive 1 charge to this nitrogen. We subtract the base state valence of nitrogen, which is 5, from 4, and so the formal charge of nitrogen bonded to 4 different atoms, is a +1. We count the formal charge valence by counting the electrons covalently bonded to nitrogen, but counting a bond as 1 electron and not two, so in this case the Nitrogen has 4 electrons. Now lets use an example where Nitrogen is bonded to 4 different atoms. The Formal charge of nitrogen bonded to 3 seperate atoms is 0, or nuetral. We take the amount of electrons in the BASE valence (5) and subtract this number by the amount of electrons we counted in the bonded atom (in our example, this was 5). We then take the base valence of Nitrogen, which is 5 valence electrons (it needs 3 to gain 8). this is a total of 5 electrons (in an atom with a valence of 8, so now you see where the difference can start to arise). Compared to our previous case, Nitrogen bonded to 3 atoms, we count 1, 2, 3 electrons (from the covalent bonds) and 2 electrons from the nitrogens lone pair. In calculating the Formal charge of an atom in a bond however, what we do is count the covalent bonds in the atom as 1 electron instead of two, and count each electron in a lone pair as a seperate electron. When counting the valence of an atom in a bond, we normally count each covalent bond in pairs of two - so if you look at nitrogen bonded to 3 things, you count each of the 3 bonds as two electrons, and the last two to get to 8 actually hang out as a lone pair. 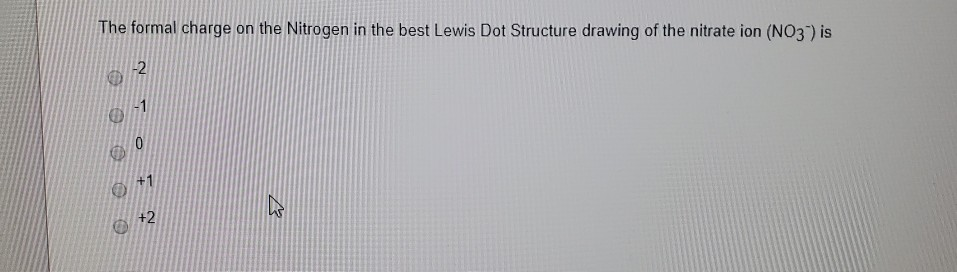 
In the case nitrogen creates 3 different bonds, it will then have a total of 8 valence electrons. Nitrogen has 5 valence electrons, needing 3 more to form an Octet. In other words, take the base state of an element and its valence. The formal charge is compairing the 'nuetral' state of an atom to the bonded state - in terms of electrons in the valence. In bond forming, we usually denote the formal charge of an atom in relation to its electrons within a bond. So an Oxygen will end up carrying an extra (-)charge on 2 out of 3 nights. The Oxygens all share the duty of taking care of the kids without bed though. So 2 of the oxygen molecules will be negatively charged. One Oxygen already has an extra kid sleeping with no bed (came from Hydrogen). So it always ends up with a positive charge. It boils down to Nitrogen never having more than 4 kids sleeping in the house. Now as a rule, all the parents are happiest when they have 8 total kids (Except Hydrogen who can only handle 2 and doesn't live here regardless). Except 1 Oxygen also brings an extra kid from it's short marriage with a Hydrogen (who is now childless). But they have a strange parenting agreement. The shared children are supposed to send half the time with each parent. And bonds between Nitrogen and Oxygen are the rooms where the shared children can sleep. The protons are like beds, The electrons are like kids. ELI5? Nitrate is a weird household of atoms. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
